*EMBARGOED All research presented at the 2022 ACG Annual Scientific Meeting and Postgraduate Course is strictly embargoed until Sunday, October 23, 2022, at 12:00 pm EDT.

Wednesday, October 26, 2022 | 9:50 AM – 10:00 AM ET | Location: Hall C2
Author Insight from Sahil Khanna, MBBS, MS, FACG, Mayo Clinic
What’s new here and important for clinicians?
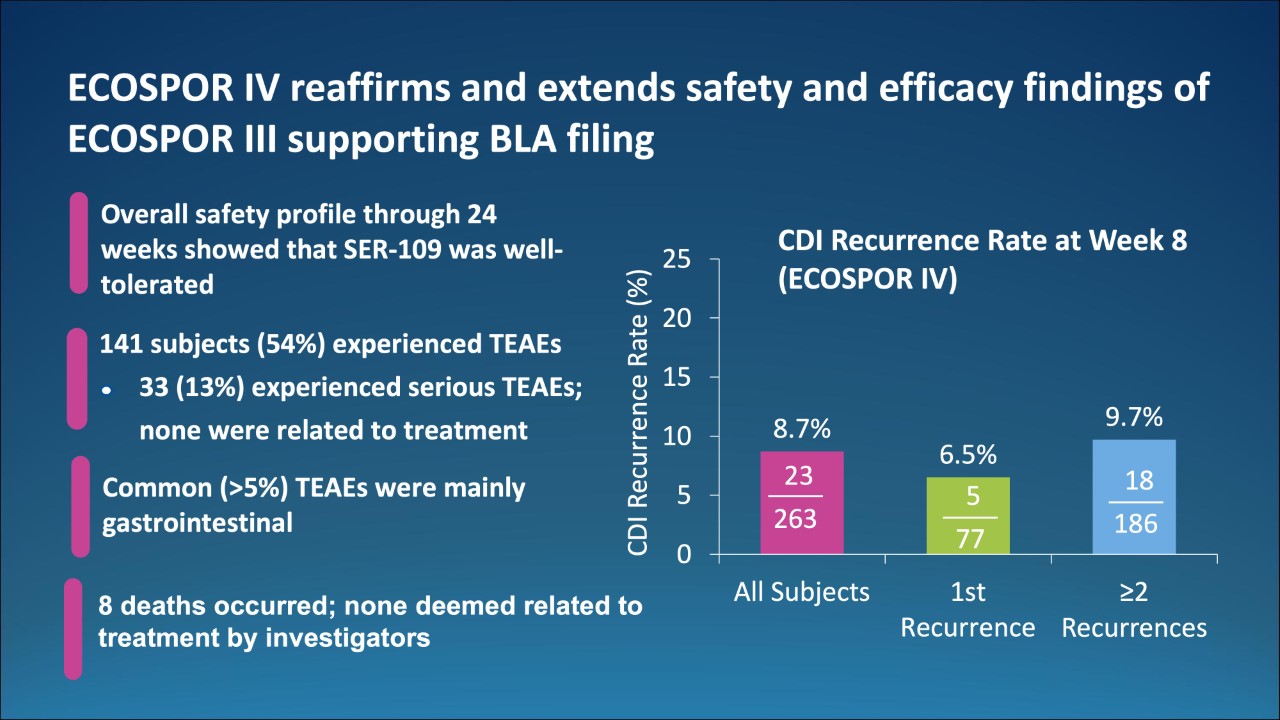
- In this open-label trial (ECOSPOR IV), the investigational microbiome therapeutic, SER-109, was well tolerated in patients with multiple comorbidities.
- The rate of recurrent Clostridioides difficile infection (rCDI) was low through Week 8, regardless of the number of prior episodes or antibiotic regimen used to treat the qualifying episode.
- These data expand upon the findings of favorable tolerability and clinical benefit of reduced risk of CDI recurrence observed in the placebo-controlled, randomized trial (ECOSPOR III) in an expanded patient population.
- The Phase 3 data support the potential benefit of SER-109 following antibiotics to prevent recurrence of this debilitating disease.
What do patients need to know?
- What is SER-109? SER-109 is a donor-derived investigational microbiome therapeutic designed to prevent recurrence of Clostridioides difficile infection (CDI) following antibiotic treatment for CDI in adults with history of recurrence.
- How is SER-109 manufactured? The SER-109 manufacturing purification process is designed to remove unwanted microbes and reduce the risk of pathogen transmission beyond donor screening alone.
- How is SER-109 administered? SER-109 is administered as 4 capsules taken orally once daily on an empty stomach for 3 consecutive days.
- What are the key clinical findings? Data from clinical studies support the potential benefit of SER-109 to prevent CDI recurrence. In a double-blind, placebo-controlled trial, 79% of patients who received SER-109 were free of CDI recurrence up to 24 weeks after treatment compared with 53% of patients who received placebo. In the open-label trial where all patients received SER-109, 86% of patients were free of CDI recurrence up to 24 weeks after treatment. SER-109 has an observed well-tolerated safety profile.
Author Contact
Sahil Khanna, MBBS, MS, FACG, Mayo Clinic
Khanna.Sahil [at] mayo.edu
Media Interview Requests
To arrange an interview with any ACG experts or abstract authors, please contact Becky Abel of ACG via email at mediaonly [at] gi.org or by phone at 301-263-9000.

