Continued Treatment With Tirzepatide Is Necessary to Maintain Weight Loss
Philip Schoenfeld, MD, MSEd, MSc (Epi)
Chief (Emeritus), Gastroenterology Section, John D. Dingell VA Medical Center, Detroit, MI.
This summary reviews Aronne LJ, Horn DB, Bays HE, et al. Continued treatment with tirzepatide for maintenance of weight reduction in adults with obesity: The SURMOUNT-4 randomized clinical trial. JAMA 2024; 3310: 38-48.
Access the article through PubMed
Correspondence to Philip Schoenfeld, MD, MSEd, MSc. Editor-in-Chief. Email: EBGI@gi.org
STRUCTURED ABSTRACT
Question: Is continued treatment with tirzepatide (Zepbound; Eli Lilly, Indianapolis, IN), a glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist, necessary for maintenance of weight loss?
Design: Thirty-six week, open-label lead-in treatment with tirzepatide, followed by 52-week, randomized, double-blind, placebo-controlled withdrawal trial.
Setting: Seventy sites in Argentina, Brazil, Taiwan, and the United States.
Patients: Eligible patients were: (a) >18 years old; (b) obesity defined as body mass index (BMI) >30; or, (c) overweight defined as BMI > 27 plus at least 1 weight-related complication (e.g., obstructive sleep apnea, hypertension, dyslipidemia, cardiovascular disease). Key exclusion criteria were diabetes and prior surgery for obesity.
Intervention: During a 36-week, open-label, treatment lead-in period, study patients were started on tirzepatide 2.5mg subcutaneous (subq) weekly and had their dose increased every 4 weeks until a maximum tolerated dose of 10 mg or 15 mg weekly was achieved. Patients also received nutritional counseling to adhere to a healthy 500 kcal/day diet and lifestyle counseling to achieve >150 minutes (2.5 hours) of physical activity per week. Study patients who achieved maximum tolerated dose of 10 mg or 15 mg tirzepatide weekly by week 36 were then randomized 1:1 to continue tirzepatide or receive matching placebo subq injections for 52 weeks.
Outcome: Primary outcome was percent change in body weight from time of randomization (week 36) through end of study at week 88, (52 weeks after randomization). Key secondary endpoints included proportion of patients maintaining >80% of weight loss from week 36 to week 88.
Data Analysis: Intention-to-treat analysis using 2-sample t test for primary outcome.
Funding: Eli Lilly, manufacturer of tirzepatide, designed and oversaw the study including data collation and analysis.
Results: Of 783 individuals who started the 36 week, open-label, lead-in treatment period, approximately 7% (n = 53) withdrew due to side effects, while 670 achieved maximum tolerated dose of 15 mg subq weekly (93%) or 10 mg subq weekly (7%). Among the 670 study patients, mean age was 48 years old, 71% female, 80% White, and mean baseline weight was 107 kg/235 pounds with mean BMI of 38.4. During the 36-week, open-label treatment period, study patients achieved mean weight loss of 21%, or approximately 22.5kg/50 pounds.
During the 52-week, randomized withdrawal period (week 36 through week 88), study patients who continued on tirzepatide had an additional mean weight loss of 5.5%, or approximately 6kg/13 pounds. However, study patients randomized to placebo subq injections regained 14.0% of body weight, or approximately 15kg/33 pounds (Figure 1). At week 88, significantly more patients treated with tirzepatide maintained at least 80% of weight loss from initial open-label treatment period compared to patients switched to placebo: 89.5% vs 16.6%, P < 0.001.
During the 36-week, open-label treatment period, 81.0% of individuals reported adverse events with gastrointestinal (GI) side effects being most common, including nausea (35.5%), diarrhea (21.1%), constipation (20.7%) and vomiting (16.4%).
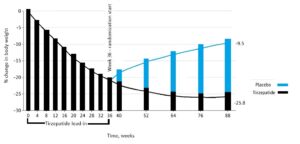 Figure 1. Tirzepatide vs placebo and body weight change. Patients were randomized at week 36.
Figure 1. Tirzepatide vs placebo and body weight change. Patients were randomized at week 36.
COMMENTARY
Why Is This Important?
When patients initiate treatment with GLP-1 receptor agonists, like semaglutide, or GIP and GLP-1 receptor agonists, like tirzepatide, they frequently ask physicians if they will need to continue the medication indefinitely in order to maintain weight loss. These data clearly demonstrate that continued medication use is necessary for the vast majority of patients.
This shouldn’t be surprising. Obesity is increasingly viewed as a chronic disease, and only bariatric surgery, endoscopic sleeve gastroplasty, and GLP-1 receptor agonist agents have demonstrated efficacy for sustained, clinically important weight loss.1-3 Unfortunately, intensive lifestyle and nutritional interventions, including restricted eating schedules, have not demonstrated similar sustained benefits.4
Hepatologists are increasingly using these agents for metabolic dysfunction-associated steatohepatitis patients with obesity, and gastroenterologists frequently see patients with GI side effects, like nausea or constipation, after starting GLP-1 receptor agonists. Therefore, we need to understand the risks and benefits of these medications as well as understanding how to mitigate side effects.
Key Study Findings
During the 36-week, open label tirzepatide treatment period, mean weight loss was 21%, or approximately 22.5 kg/50 pounds.
Caution
GI side effect were common during the 36-week, open-label treatment period, and included nausea (35.5%), diarrhea (21.1%), constipation (20.7%), and vomiting (16.4%). Although tirzepatide was recently approved by the US Food and Drug Administration for obesity, lack of insurance coverage and high out-of-pocket costs remain potential barriers to maintenance use.
My Practice
Since I’m not an obesity specialist, I consulted with one of our former Associate Editors, Sonali Paul, MD, MS, who is certified in obesity medicine and has expertise in using these medications for management of obese metabolic dysfunction steatohepatitis patients. She noted the following pearls for management, which we have discussed in prior EBGI summaries.1,4-5
When prescribing GLP-1 receptor agonists, the dose should be gradually increased in 2.5 mg increments every 4 weeks based on tolerability. Treatment should be reverted to a lower dose if clinically important nausea develops. If patients develop mild constipation, treatment with an osmotic laxative without lowering the dose is acceptable. Continued treatment will be required for maintenance of weight loss in the majority of patients since obesity is a chronic disease, although the lowest effective dose should be used.
There does appear to be a small risk of developing pancreatitis based on all available data,5 so do not use in patients with a history of pancreatitis. As discussed in a prior EBGI summary,5 significant weight loss does increase the risk of gallstone development and also may increase the risk of cholecystitis and choledocholithiasis. Current data is insufficient to support a causal link between GLP-1 receptor agonists and gastroparesis or bowel obstruction.5
Finally, as discussed in a prior EBGI summary,5 whether or not GLP-1 receptor agonists need to be discontinued prior to endoscopic procedures to minimize aspiration risk during monitored anesthesia care remains controversial. Although the American Society for Anesthesiology updated their pre-operative fasting guidelines in 2023 and recommended that subq injections of GLP-1 receptor agonists should be held for 1 week, there is insufficient research data to support this recommendation and position statements from our GI societies do not support this. Nevertheless, many endoscopists and patients will be required by their anesthesiology team to hold subq injections of GLP-1 receptor agonists for one week if deep sedation with propafol is used. In my own practice, I do not routinely hold GLP-1 receptor agonists when performing colonoscopy or even upper endoscopy with midazolam and fentanyl for sedation.
For Future Research
Ongoing research will investigate other potential long-term adverse events that could be associated with weight loss, including sarcopenia.
Conflict of Interest
Dr. Schoenfeld has no relevant conflicts of interest.
REFERENCES
- Paul S. Tirzepatide for obesity: “Mounting” evidence for substantial weight loss. Evidence-Based GI; Aug 2022:11-15. https://gi.org/journals-publications/ebgi/paul_august_2022/
- Kolb J, Chiang A. Endoscopic sleeve gastroplasty is effective for patients with obesity who MERIT intervention. Evidence-Based GI; Feb 2023:6-11. https://gi.org/journals-publications/ebgi/kolb_chiang_feb2023/
- Fu Y, Paul S. Bariatric surgery is superior to lifestyle changes + best medical care for metabolic dysfunction-Associated steatohepatitis: The BRAVES study. Evidence-Based GI; July 2023: 11-15. https://gi.org/journals-publications/ebgi/fu_paul_july2023/
- Paul S. Effectiveness of intermittent fasting for weight loss: It’s not just when you eat, but what you eat, too! Evidence-Based GI; Feb 2022:13-16. https://gi.org/journals-publications/ebgi/paul_february2022/
- Schoenfeld P, Paul S. GI adverse events with GLP-1 receptor agonists for weight loss: Understanding the risks. Evidence-Based GI: Nov 2023:18-21. https://gi.org/journals-publications/ebgi/phil_paul_nov2023/


